Schedule a Call Back
- Agricultural Machinery & Accessories
- Building & Construction Machinery & Accessories
- Building, Construction and Maintenance Services
- Crushing, Granulating and Size Reduction Machines
- Meteorological and Land Surveying Instruments
- Mining and Oil Refining Machinery & Accessories
- Wood and Stone Working Machinery
- Discrete Semiconductor Devices
- Electric Power Generation and Related Equipment
- Electric Traction and Moving Systems
- Electrical Boxes and Enclosures
- Electrical Control Devices
- Electrical Lugs, Plugs and Connectors
- Electrical Measuring and Testing Instruments
- Electrical Systems, Components & Accessories
- Electronic Control Devices
- Electronic Systems, Components & Accessories
- Fuse, Circuit Breakers, Switchgears and Protective Devices
- Printed Circuits and Integrated Circuits
- Resistors, Capacitors and Inductors
- Transformers and Power Conditioning Equipment
- Winding and Wire Processing Machines
- Switches and Relays
- Audio and Visual Presentation Equipment
- Computer Hardware, Software & Accessories
- Consumer Electronics
- Data Management Systems
- Doors, Windows and Hardware
- Displays, Signage and Accessories
- Finished Ferrous, Non-Ferrous and Non Metallic products
- Gauges and Fixtures
- Housings, Cabinets and Casings
- Industrial Chemicals, Compounds and Mixtures
- Industrial Services
- Lamps, Lighting Systems & Accessories
- Lasers and Laser Equipment
- Leather Machinery & Accessories
- Light, Wave and Sound Generating and Measuring Instruments
- Machinery Components & Accessories
- Magnetic and Non-Magnetic Materials
- Medical Equipment and Accessories
- Minerals, Ores, Metals and Alloys
- Miscellaneous Industrial Supplies
- Office Equipment and Supplies
- Optics, Optical Machinery and Equipment
- Sheet Metal Components, Precision and Turned Components
- Textile and Fabric Machinery & Accessories
- Transportation, Storage and Mail Services
- Cutting Tools & Accessories
- Cutting Machinery & Accessories
- Grinding Machines & Accessories
- Hand Tools, Power Tools & Accessories
- Lathes and Turning Centres
- Machine Tools & Accessories
- Machining Centres
- Metal Forming Machinery & Accessories
- Metal Treatment Machinery & Accessories
- Milling and Bending Machines
- Power Tools & Accessories
- Presses and Press Brakes
- Special Purpose Machines
- Pumps, Pump Parts & Accessories
- Valves, Valve Components & Accessories
- Abrasive Handling Pumps
- Submersible Pumps
- Centrifugal Pumps
- Solar Water Pumps
- Borewell Pumps
- Pressure Pumps
- Axial Flow Pumps
- Barrel Pumps
- Booster Pumps
- Borewell Submersible Pumps
- Cantilever Pumps
- Chemical Pumps
- Condensate Pumps
- Dewatering Pumps
- Diaphragm Pumps
- Dosing Pumps
- Drum Pumps
- Fluid Transfer Pumps
- Foot Pumps
- Fountain Pumps
- Gas Seal Slurry Pumps
- Gear Pumps
- Grinder Pumps
- Hand Pumps
- High Temparature Pump
- Highly Volatile Liquid Pumps
- Hydraulic Pumps
- Injection Pumps
- Inline Pumps
- Jet Pumps
- Lobe Pumps
- Metering Pumps
- Monoblock Pumps
- Mud Pumps
- Multistage Pumps
- Openwell Pumps
- Refrigerators Pump
- Rotary Gear Pump
- Screw Pump
- Seawater Pumps
- Self Priming Pumps
- Sewage Pumps
- Slurry Pump
- Split Casing Pumps
- Suction Pumps
- Sump Pumps
- Syringe Pumps
- Turbine Pumps
- Vacuum Pumps
- Drainage Pumps
- Calibration, Inspection and Viewing Instruments
- Dimension & Weight Measuring Instruments
- Electrochemical and Chromatographic Measuring Instruments
- Electronic and Communication Measuring & Testing Instruments
- Flow & Level Measuring and Control Instruments
- Indicating and Recording Instruments
- Mechanical Strength, Metal and Structural Testing Instruments
- Meters
- Power Analysers, Timers and Counters
- Pressure Measuring and Control Instruments
- Solid-Liquid-Gas Analysers and Monitors
- Temperature Control and Measuring Equipment
- Tension Measurement and Control
- Testing and Measuring Instruments
- Visual Inspection Equipment
- Analysers, Analysis Systems and Monitors
IPF NEWSLETTER
Subscribe to our newsletter and
never miss a story

search Result
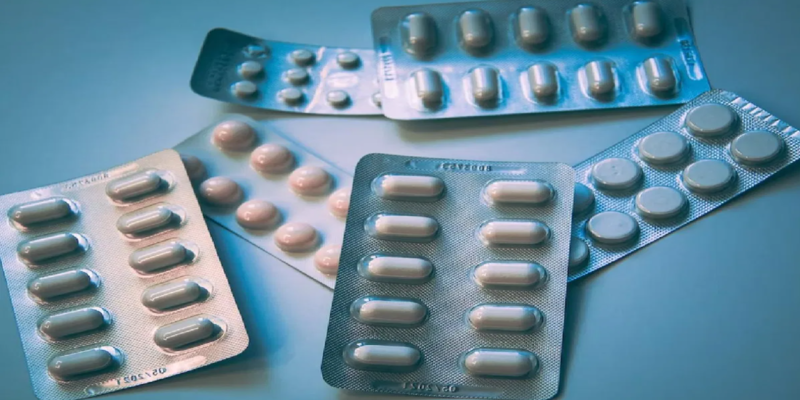
Union Health Ministry enhances Pharma Quality Control with Revised Schedule M rules
Schedule M outlines the Good Manufacturing Practices (GMP) for pharmaceutical products, and the revised version aims to ensure adherence to GMP standards, emphasising the requirements for premises, plant, and equipment related to pharmaceutical production.Read more

India marks Oct 10 as inaugural cGMP day to elevate pharmaceutical standards
The government has made it obligatory for all drug manufacturers to adhere to Good Manufacturing Practice (GMP) guidelines.Read more












